This isn't really a library entry, but I couldn't think of a better place to put it. I'm sorry! 
Ohko's Guide to Semi-Realistic Bronze Age Smithing
For Dummies (and Authors)
Introduction
So you want your character to do smithing? But you have no idea where to start? This guide provides an overview of some methods of metalworking used on Earth. Feel free to use this as a reference for your writing... or not!

Adventurer's Guild is a fantasy roleplaying game, and there's no need to be realistic.
However, if you want to add a touch of realism to your writing, I hope this resource can help! While I'm not an expert on this subject by any means, I'm a total geek on primitive technology — so take whatever you like from this!
Ores, Metals, and Alloys
Of these seven metals, only gold and silver are found in their native (elemental) form. The remainder are all usually found as rock-like ores, which mean that they are chemically fused to another element (e.g. iron oxide). Ores need to be smelted in order to purify them into their native (elemental) form.
While native metals are great, they aren't always particularly useful for a metalworker trying to forge armor, swords, or tools. All of the native metals (except iron) are generally quite soft and easy to melt in a fire. Think of copper wire, tin cans, gold foil... they are easily bent and useless as a weapon.
In order to change the properties of metals, they are often mixed with something else to create an alloy. Alloys can have have different useful properties such as increased stiffness, flexibility, density, strength, or even rust resistance.
Where to Find Ores?
| Iron | Copper | Lead | Tin | Silver | Mercury | Gold |
|---|
| 4.1% Earth's Mass | 50 ppm | 14 ppm | 2.2 ppm | 0.07 ppm | 0.05 ppm | 0.001 ppm |
| Grey / Red / Brown | Green / Grey | Grey / Black / Silver | Black / Grey | Grey / Silver | Bright Red | Gold |
| 1538 °C | 1084 °C | 327 °C | 231 °C | 961 °C | -39 °C | 1064 °C |
In order to understand where to find ores, it is extremely helpful to know the relative abundance of different metals.
Iron ore is extremely common. In fact, any kind of slightly reddish rock probably has some iron in it. This probably means that your backyard has iron! The question is, how much? Do the rocks in your backyard have 10% iron? 15% iron? Should you call a mining company and get rich???
For practical reasons, we call something an "ore deposit" if the rocks in the area contain enough metal content for it to be profitably extracted. In the modern day, we have a lot of fancy tools that allow us to calculate the metal content of various rock. As a result, on Earth we often see mining companies set up shop in areas where they're drilling straight into ground to mine tons of plain-looking grey rock...
However, in the ancient days, humans didn't have access to fancy tools. People depended on the color of rocks (and other things) in order to identify ore. It was easiest to see bands of rocks (an "ore vein") in sedimentary layers of the earth that were displaced, such as on cliff-sides or exposed mountains. If humans spotted an unusual color, perhaps there was metal ore there!
Fortunately, a few varieties of ores have a very distinctive color. One unique variety of copper ore — Malachite — has a glaring green color. Of course, there are many other types of (greyish) copper ore, but Malachite was likely the earliest variety discovered by humans due to its ease of identification. If you were a prehistoric Assyrian shepherd and walking by a cliff, and you spotted streaks of green on the cliffside... it was likely there was copper ore here! We call those streaks of green an "ore vein". The shepherd would go call his miner friends and they would chip away at this "surface vein". | |
Once the original surface vein was discovered, it's only the tip of the iceberg!
Much of the vein continues underground! |
If you have good mining skills, you can start digging shafts to follow the veins. Miners do not dig randomly. They follow the color of their rock. |
A Brief History of Early Metals, Copper, and Bronze
Due to the distinctive color of malachite, copper was probably one of the earliest metals to be worked by humans. Very few other ores/metals (except cinnabar and gold) have such a distinctive color, so they could have only been discovered by blind accident.
Fortunately, most of the metals of antiquity (except iron) have a low melting point.
Lead and Tin are excellent examples. Both ores have low enough melting points (327 °C) (231 °C) that they can be smelted in an ordinary campfire. In fact, this was likely the way these metals were discovered. Someone left a bunch of grayish rocks in the village campfire, and a couple hours later they saw that the rocks had melted into this shiny stuff. Weird!
So how do you smelt lead and tin? It's super easy! You just melt it! Both lead and tin have a melting point lower than typical rocks. As a result, if you heated a bunch of ore (rocks) in a spoon, the lead/tin would turn liquid. Any unmelted rock would stay at the bottom of the ladle, and you could pour the pure metal off the side. Typically, the molten lead/tin would be poured into a mold. When the metal solidified, it would take the shape of the mold. This method of metalworking is called "casting". It was possible to make many intricate shapes with metal casting. For instance, lead balls were created by pouring molten lead into spherical molds. | |
Smelting copper is a bit more challenging with its higher melting point (1084 °C). Copper, Silver, and Gold could not be smelted in an ordinary campfire (max campfire temperature ~900 °C). Instead, humans needed to invent a new method to raise the temperature higher enough to melt these metals.
There are three simple ways to raise the temperature of a fire: For an improved fuel, charcoal was used. Charcoal burns hotter (and cleaner) than wood. Charcoal is made from wood burned in the absence of oxygen, which causes it to retain all its energy. To increase oxygen, humans invented bellows, which could blow air at a fire. It is extremely labor-intensive to maintain the bellows for a furnace, which often needed to go nonstop for hours/days. Slowing down or speeding up will immediately change the temperature of the fire. It was common for a team of multiple individuals to focus on the bellows. An ancient copper smelter required at least 4-6 people simultaneously trying to blow oxygen into the fire pit. To conserve heat, pits were dug into the ground. The earth could insulate against heat escaping. Clay walls were built to capture heat. Finally, a crucible containing ore was placed in the center of the furnace amidst a ton of charcoal. When the process was complete, it would result in molten metal that could be cast into a mold. | |
The problem with a metal like copper is that it is simply too soft. Although copper was abundant (and shiny and pretty), it was not practical for making any tools by itself.
To harden copper into something stiffer, humans discovered a few copper alloys:
- 88% Copper + 12% Arsenic = Bronze
- 88% Copper + 12% Tin = Bronze
- 65% Copper + 35% Zinc = Brass
All of these copper alloys are predominantly copper (~90%) plus a trace amount of a stiffening metal. Increasing the fraction of the stiffening metal generally makes the resultant metal harder (yet also more brittle). For example, raising the concentration of tin in bronze results in a harder/sharper sword, but it is more likely to shatter.
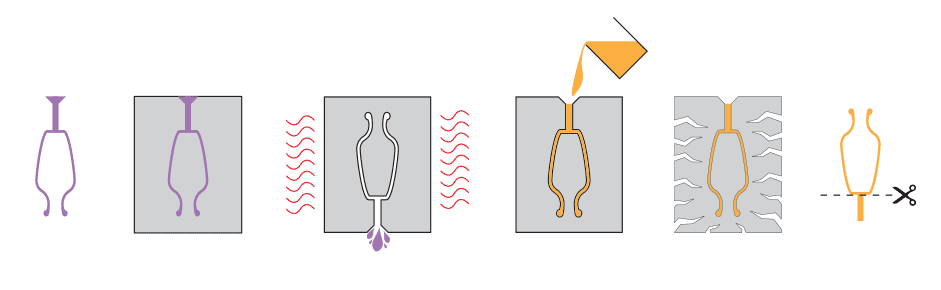
Casting bronze is fairly simple. By heating the appropriate proportions of copper and alloy in a crucible, molten bronze was created. Molten bronze would then be poured into a mold, typically made out of packed sand or clay. Advanced shapes could be created using fancy methods like Lost Wax Casting. This method entailed (1) creating a shape out of beeswax, (2) using the beeswax as a template in the mold, (3) melting the beeswax, and (4) pouring the bronze.
Generally, bronze age tools and objects were all creating by casting, not hammering (like iron).
After casting a tool, it would often be "finished" with extensive amounts of grinding, sanding, and other final touches. Bronze can be "work-hardened" (e.g. hammering) to a limited extent, but it is much more brittle than iron. If pushed too hard, it will shatter. Bronze swords were known to shatter on the battlefield in antiquity. This was in contrast with iron/steel swords which were less likely to shatter.
It should be noted that bronze swords were often regarded as superior to wrought iron swords particularly in the Early-Middle Iron Age. The quality of wrought iron was highly variable in the early period and often full of impurities. Bronze often produced higher quality products and high-tin alloys were a harder metal than wrought iron. Additionally, bronze does not rust.
The Iron Age eventually overtook the Bronze Age primarily economic reasons.
The primary tin ore of antiquity was Cassiterite, which has a distinctive black crystalline appearance. However, it was extremely rare and found in only a handful of regions across the world. Notably, the entirety of the Middle East lacked a major source of tin.
This is ironic because the most Bronze Age civilizations: Anatolia (Turkey), Egypt, Mesopotamia, Greece, India, and Central Asia all lacked their own regional sources of tin. Trade with faraway countries was critical for the bronze economy, which meant that a handful of powerful countries monopolized bronze production. When trade routes shut down, military economies were paralyzed and countries were left extremely vulnerable to their enemies.
Iron — in contrast — was everywhere (4% of the Earth's surface). When metallurgy technology advanced to cover iron, the democratization of military weapons resulted in regional upheaval that drove the Late Bronze Age collapse. Suddenly, formerly "irrelevant" barbarians and raiders in backwater countries could field armies of massed infantry larger than ever before, overwhelming the smaller elite forces of Bronze Age charioteer nobility.
| Map of Known Tin Sources in Antiquity
|
Epilogue
In conclusion, this article summarized basics of metallurgy relevant to the Bronze Age period.
I'm tired for today, so I will stop here. I hope people did not find this excessively dull or useless.
Next time, I will pick up with Iron Age metallurgy~